|
Unfortunately, elemental phosphorus is volatile and highly toxic. Early matches were pieces of wood coated with elemental phosphorus that were stored in an evacuated glass tube and ignited when the tube was broken (which could cause unfortunate accidents if the matches were kept in a pocket!). During the 19th century, the demand for phosphorus for matches was so great that battlefields and paupers’ graveyards were systematically scavenged for bones. For more than a century, the only way to obtain phosphorus was the distillation of urine, but in 1769 it was discovered that phosphorus could be obtained more easily from bones. Non-metals Period - reactivity increases as you go from the left to the right across a period. (Unfortunately for Brandt, however, it did not turn lead into gold.) The element was given its current name (from the Greek phos, meaning “light,” and phoros, meaning “bringing”) in the 17th century. Group - reactivity increases as you go down a group The farther to the left and down the periodic chart you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity. Oxygen, at the top of group 16 (6A), is a colourless gas in the middle of the group, selenium is a semiconducting solid and, toward the bottom, polonium is a. For example, it glowed in the dark and burst into flames when removed from the water. The urine was distilled to dryness at high temperature and then condensed the last fumes were collected under water, giving a waxy white solid that had unusual properties. Believing that human urine was the source of the key ingredient, Brandt obtained several dozen buckets of urine, which he allowed to putrefy. 
Phosphorus was discovered in 1669 by the German alchemist Hennig Brandt, who was looking for the “philosophers’ stone,” a mythical substance capable of converting base metals to silver or gold. Consequently, its alloys expand as they cool, filling a mold completely and producing crisp, clear letters for typesetting. Bismuth is used in printing because it is one of the few substances known whose solid state is less dense than the liquid. Its name comes from the old German wismut, meaning “white metal.” Bismuth was finally isolated in the 15th century, and it was used to make movable type for printing shortly after the invention of the Gutenberg printing process in 1440. The history of bismuth (Bi), in contrast, is more difficult to follow because early alchemists often confused it with other metals, such as lead, tin, antimony, and even silver (due to its slightly pinkish-white luster). In the form of its yellow sulfide ore, orpiment (As 2S 3), arsenic (As) has been known to physicians and professional assassins since ancient Greece, although elemental arsenic was not isolated until centuries later. Small vases of ground stibnite have been found among the funeral goods buried with Egyptian pharaohs. (b) A fragment of an Egyptian painting on limestone from the 16th–13th centuries BC shows the use of ground stibnite (“kohl”) as black eye shadow. (a) Crystals of the soft black mineral stibnite (Sb 2S 3) on a white mineral matrix. Group - reactivity increases as you go down a group. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.\): The Ancient Egyptians Used Finely Ground Antimony Sulfide for Eye Makeup. In Metals: Period - reactivity decreases as you go from left to right. 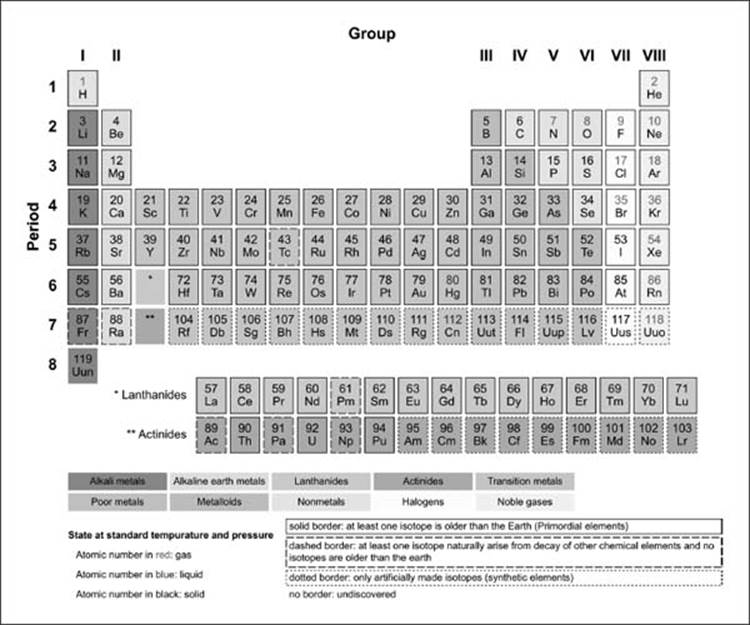
Because electrons are more easily supplied or taken away as you move farther left and down the periodic chart, reactivity increases as you move further left and down the periodic chart. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). A Simple Guide to the Trends of Chemical Reactivity in Periodic Table Period - As you move from left to right over a period, your reactivity reduces as well. Free Gift for you: Interactive Periodic Table 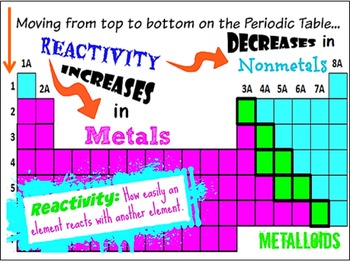 
In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
